|
1/13/2023 0 Comments Atomic orbitals explained 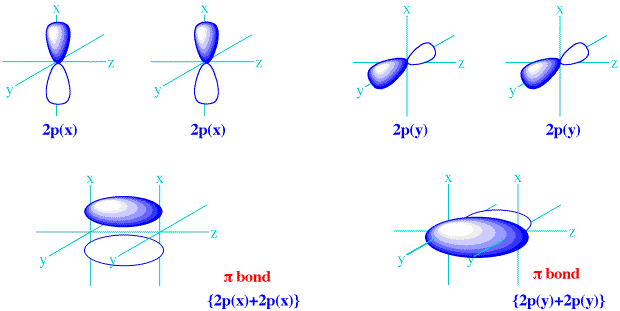
Moreover, it sometimes happens that the configuration interaction expansion converges very slowly and that one cannot speak about simple one-determinantal wave function at all. Nevertheless one has to keep in mind that electrons are fermions ruled by Pauli exclusion principle and cannot be distinguished from the other electrons in the atom. For example, one can say for a given transition that it corresponds to the excitation of an electron from an occupied orbital to a given unoccupied orbital. The atomic orbital concept is therefore a key concept for visualizing the excitation process associated to a given transition. This notation means that the corresponding Slater determinants have a clear higher weight in the configuration interaction expansion. 1 s 2 2 s 2 2 p 6 for the ground state of neon - term symbol: 1S 0). by occupations schemes of atomic orbitals (e.g. These states are labelled by a set of quantum numbers summarized in the term symbol and usually associated to particular electron configurations, i.e. In atomic physics, the atomic spectral lines correspond to transitions (quantum leaps) between quantum states of an atom. (When one considers also their spin component, one speaks of atomic spin orbitals.) The spatial components of these one-electron functions are called atomic orbitals. the eigenstates of the atomic Hamiltonian, is expanded (see configuration interaction expansion and basis (linear algebra)) into linear combinations of anti-symmetrized products (Slater determinants) of one- electron functions. In quantum mechanics, the state of an atom, i.e. 
, the letters associated with those numbers are K, L, M, N, O. In some cases, the principal quantum number is given a letter associated with it. Where X is the energy level corresponding to the principal quantum number n, type is a lower-case letter denoting the shape or subshell of the orbital and it corresponds to the angular quantum number l, and y is the number of electrons in that orbital.įor example, the orbital 1 s 2 ( pronounced "one ess two") has two electrons and is the lowest energy level ( n = 1) and has an angular quantum number of l = 0.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
